In today’s rapidly evolving world of health and wellness, an intriguing concept is capturing the attention of both scientists and the health-conscious public alike: Biological Age Testing. This revolutionary approach to understanding our bodies goes beyond the traditional markers of health, offering a more nuanced view of aging and well-being. But what exactly is a biological age test, and more importantly, what can it do for you?
Biological age refers to the condition of our bodies relative to our chronological age, the simple count of years since birth. While two individuals may share the same chronological age, their biological ages can differ significantly, influenced by a myriad of factors including genetics, lifestyle choices, environmental exposures, and more. This disparity opens up a fascinating window into personalised health, suggesting that the aging process is more malleable than previously thought.
Through Biological Age Testing, which often involves sophisticated analyses like DNA methylation patterns, telomere length measurement, or advanced biomarker profiling, individuals can gain insights into their true biological condition. This information is not just a number but a comprehensive assessment of one’s health, potentially revealing the early signs of aging-related diseases, lifestyle impacts, and even the effectiveness of anti-aging interventions.
In this blog, we will dive deep into the world of Biological Age Testing, exploring its mechanisms, the science behind it, and how it stands to revolutionize personal and public health strategies. From understanding how biological age is determined to uncovering actionable steps you can take to potentially ‘turn back the clock’, we aim to provide a thorough overview of what biological age testing can do for you. Whether you’re a health enthusiast seeking to optimize your wellness journey, or simply curious about the aging process, join us as we unravel the mysteries of biological age and its profound implications for our lives.
Epigenetic clock theory of aging cell
The Epigenetic Clock Theory of Aging posits that the accumulation of epigenetic changes, particularly DNA methylation alterations, is a key driver of the aging process. This theory is grounded in the observation that specific patterns of DNA methylation, which are modifications that affect gene expression without altering the DNA sequence, change predictably as an individual ages. These patterns can be quantified to create an “epigenetic clock,” a biomarker that predicts biological age with high accuracy across different tissues and cell types.
Foundations of the Epigenetic Clock
The epigenetic clock is based on the methylation status of specific cytosine-phosphate-guanine (CpG) sites in the genome. Methylation patterns at these sites change in a predictable manner with age, and by analyzing these patterns using sophisticated algorithms, scientists can estimate an individual’s biological age. This age may differ from their chronological age, providing insights into their biological condition, healthspan, and lifespan.
Key Aspects of the Theory
1. Predictive Power for Aging and Health: The epigenetic clock has been shown to predict lifespan and healthspan more accurately than chronological age. Individuals with an accelerated epigenetic age compared to their chronological age are at a higher risk for aging-related diseases and mortality.
2. Universal Aging Process: The consistency of epigenetic changes across different types of tissues suggests a universal aging mechanism. This universality supports the idea that the epigenetic clock captures a fundamental aspect of the biological aging process.
3. Interventional Potential: Since epigenetic modifications are reversible, unlike DNA sequence changes, the epigenetic clock theory opens the door to potential interventions that could slow aging or reverse age-related changes in the epigenome.
Implications of the Theory
– Aging Research: The epigenetic clock has become a vital tool in aging research, enabling the study of the aging process in unprecedented detail. It allows researchers to evaluate the efficacy of anti-aging interventions in a quantifiable manner.
– Disease Prediction and Prevention: By providing a more accurate measure of biological aging, the epigenetic clock can help predict the risk of developing age-related diseases before they manifest, offering opportunities for early intervention and prevention.
– Understanding the Biology of Aging: The theory enhances our understanding of how epigenetic factors contribute to aging and the variability in how individuals age. It suggests that managing epigenetic health could be key to extending healthspan and lifespan.
Challenges and Future Directions
– Causal Relationship: Determining whether changes in DNA methylation are a cause or consequence of aging remains a challenge. Ongoing research aims to elucidate the causal relationships between epigenetic alterations and the aging process.
– Mechanistic Insights: While the epigenetic clock provides a powerful tool for measuring biological age, the biological mechanisms that underpin the clock’s tick remain to be fully understood. Research is ongoing to identify the molecular pathways involved in the epigenetic aging clock and how they influence health and disease.
– Therapeutic Interventions: Identifying interventions that can reset or slow the epigenetic clock could have significant implications for aging and age-related disease treatment. This includes lifestyle interventions, pharmaceuticals, and other therapies aimed at modifying epigenetic marks.
Epigenetics: An accurate cellular clock
Epigenetics refers to the study of changes in gene expression that do not involve alterations to the underlying DNA sequence—a change in phenotype without a change in genotype. These changes can affect how cells read genes, turning them on or off and affecting how they function. One of the most fascinating developments in the field of epigenetics is the discovery and refinement of epigenetic clocks, which serve as a promising tool for understanding the aging process at a cellular level.
An epigenetic clock is a biochemical test that can predict the age of cells, tissues, or organisms based on the DNA methylation levels of specific genes. DNA methylation is a chemical modification to DNA that can change the activity of a DNA segment without changing the sequence. When located in a gene promoter, DNA methylation typically acts to repress gene transcription. The patterns of these methylation sites change as we age, and by analyzing these patterns, scientists can develop algorithms that accurately predict biological age, which may differ from chronological age.
The Significance of Epigenetic Clocks
Epigenetic clocks have several significant implications:
1. Aging Research: They provide a new tool for understanding the mechanisms behind aging, potentially identifying why some individuals age faster or slower than others and uncovering the relationship between genetics, lifestyle factors, and aging.
2. Disease Prediction and Prevention: Because the biological age indicated by epigenetic clocks can differ from chronological age, they may help identify increased risks for age-related diseases earlier in life, offering opportunities for preventive interventions.
3. Evaluation of Anti-aging Therapies: Epigenetic clocks can serve as a biomarker to assess the efficacy of anti-aging interventions, helping to distinguish between treatments that have genuine rejuvenating effects from those that do not.
4. Forensic Applications: Beyond health and aging, epigenetic clocks can be used in forensic science to estimate the age of unidentified individuals or to track developmental changes in tissues.
Challenges and Future Directions
Despite their potential, epigenetic clocks face challenges and limitations. One issue is the variability in methylation patterns caused by environmental factors, lifestyle choices, and underlying health conditions, which can affect the accuracy of age predictions. Additionally, while epigenetic clocks can predict age and are associated with disease risks, they do not yet fully explain the biological mechanisms driving aging or the development of age-related diseases.
Future research aims to refine these clocks to account for such variability and to understand the biological underpinnings of the methylation patterns used in age predictions. Scientists are also exploring the development of “interventional” epigenetic clocks that can more directly measure the effects of anti-aging treatments at the cellular level.
Genetic estimators in the Horvath clock
The Horvath clock is a highly recognized epigenetic clock developed by Dr. Steve Horvath, a professor of human genetics and biostatistics. It is based on the DNA methylation levels of 353 specific sites (CpG sites) in the human genome. The Horvath clock can accurately estimate the age of a wide range of tissues and cell types, reflecting the biological age of the tissue rather than its chronological age. This tool has profound implications for aging research, offering insights into the biological processes of aging and the potential to evaluate interventions aimed at slowing the aging process.
Key Features of the Horvath Clock
1. Multi-tissue Applicability: Unlike other epigenetic clocks that may only apply to specific tissues, the Horvath clock has the unique advantage of being applicable across a wide variety of tissues and cell types, including blood, brain, breast, kidney, liver, and lung. This versatility makes it a valuable tool for comprehensive studies on aging.
2. Genetic Estimators: The genetic estimators in the Horvath clock are the DNA methylation levels at 353 specific CpG sites in stem cell. DNA methylation is a form of epigenetic modification where a methyl group is added to the DNA molecule, typically acting to suppress gene expression. The pattern of methylation at these sites changes with age, and the Horvath clock uses the methylation state of these CpG sites to predict biological age. The selected CpG sites are strategically chosen for their strong correlation with age across tissues and cell types.
3. Accuracy: The Horvath clock is known for its accuracy in estimating biological age, with a median error of 3.6 years across all analyzed samples. This precision is remarkable, considering the diverse range of tissues it can accurately age-predict.
4. Biological Age vs. Chronological Age: One of the most intriguing aspects of the Horvath clock is its ability to highlight differences between biological and chronological age. Individuals whose biological age (as estimated by the horvath’s clock) is higher than their chronological age may have an increased risk of aging-associated diseases and mortality. Conversely, a younger biological age suggests a lower risk.
Implications and Applications
– Aging Research: The Horvath clock has become a critical tool in aging research, allowing scientists to explore the mechanisms of aging and identify potential biomarkers for age-related diseases.
– Lifestyle and Intervention Studies: It can be used to evaluate the effect of lifestyle factors, dietary interventions, and drugs on the aging process, offering a quantitative measure of their impact on biological aging.
– Disease Prediction and Prevention: The clock’s ability to estimate biological age can help predict the onset of age-related diseases before they manifest, offering a window for early intervention.
Despite its widespread use and potential, the Horvath clock and other epigenetic clocks face challenges, including understanding the causal relationships between methylation changes and aging. Further research is needed to decipher whether changes in DNA methylation are a cause or a consequence of aging and how these changes relate to the biological processes underlying aging and age-related diseases.
As research advances, it is expected that epigenetic clocks will be refined and potentially combined with other biomarkers of aging to provide a more comprehensive understanding of the aging process. This could lead to the development of targeted interventions to slow aging and prevent age-associated diseases, marking a significant step forward in personalized medicine and healthy aging strategies.
Relationship to a cause of biological aging
The relationship between epigenetic changes, as measured by tools like the Horvath clock, and the cause of biological aging is an area of active and ongoing research in the field of biogerontology. It’s important to understand that aging is a complex and multifactorial process influenced by a combination of genetic, environmental, and lifestyle factors. The current understanding of how epigenetic changes relate to the cause of biological aging involves several key concepts:
1. Epigenetic Changes as a Marker of Aging: Epigenetic changes, especially DNA methylation patterns, are one of the most robust biomarkers of aging. The patterns of methylation at specific sites in the DNA change predictably as we age. Tools like the Horvath clock use these changes to estimate biological age. However, whether these epigenetic changes are a cause or a consequence of aging remains a subject of debate.
2. Epigenetic Drift and Aging: Over time, the epigenome can become dysregulated in a process known as epigenetic drift, which is characterized by changes in DNA methylation patterns. This drift may lead to altered gene expression and contribute to the aging process by affecting cellular function, increasing susceptibility to diseases, and decreasing the body’s ability to maintain homeostasis.
3. The Role of Epigenetics in Cellular Senescence: Cellular senescence is a state where cells stop dividing and undergo significant changes in gene expression. Epigenetic alterations can contribute to the induction and maintenance of senescence. Cellular senescence is considered one of the hallmarks of aging and contributes to age-related tissue dysfunction.
4. Epigenetics and Age-Related Diseases: Many age-related diseases, including cancer, Alzheimer’s disease, and cardiovascular diseases, have been linked to epigenetic changes. These changes can affect gene expression patterns critical for maintaining cellular health and function.
5. Potential Reversibility of Epigenetic Changes: One of the most intriguing aspects of epigenetic changes is their potential reversibility, which has significant implications for aging and age-related diseases. Unlike genetic mutations, epigenetic alterations can potentially be reversed, offering the possibility of therapeutic interventions that could modify the course of aging or alleviate age-related diseases.
6. Causation vs. Correlation: While there is a strong correlation between epigenetic changes and aging, proving causation is more challenging. It is unclear whether epigenetic changes drive the aging process or whether they are simply a byproduct of other underlying processes that cause aging.
Genetic studies of epigenetic age acceleration
Genetic studies of the epigenetic mechanisms of age acceleration are pivotal for unraveling the complexities of aging and the discrepancies between biological and chronological age. Epigenetic age acceleration refers to the phenomenon where an individual’s biological age, as determined by epigenetic markers, exceeds their chronological age, suggesting that their body may be aging at an accelerated pace. This area of research is crucial for identifying the genetic underpinnings of aging and potential interventions to mitigate age-related diseases and extend healthy lifespan.
Understanding Epigenetic Age Acceleration
Epigenetic age acceleration is measured using various epigenetic clocks, such as the Horvath clock or the Hannum clock, which estimate biological age based on DNA methylation patterns at specific CpG sites across the genome. Researchers compare the predicted biological age to the individual’s chronological age to determine the rate of age acceleration.
Genetic Studies and Their Findings
1. GWAS (Genome-Wide Association Studies): GWAS have been instrumental in identifying genetic variants associated with epigenetic age acceleration. These studies scan the genomes of numerous individuals to find single nucleotide polymorphisms (SNPs) that correlate with variations in the rate of epigenetic aging. For instance, GWAS have identified SNPs in or near genes like TERT (telomerase reverse transcriptase) and RBL2 (retinoblastoma-like 2) that are associated with differences in biological age.
2. Heritability of Epigenetic Age Acceleration: Studies suggest that epigenetic age acceleration has a genetic component, indicating that part of the variation in how quickly individuals age can be attributed to their genetic makeup. Heritability estimates for epigenetic age acceleration vary but demonstrate that genetics play a significant role.
3. Link to Age-Related Diseases: Genetic loci associated with epigenetic age acceleration often overlap with those implicated in age-related diseases, such as cardiovascular disease, Alzheimer’s disease, and cancer. This overlap suggests that the mechanisms driving epigenetic age acceleration may also contribute to the pathogenesis of these conditions.
4. Molecular Pathways: Research into the genetic basis of epigenetic age acceleration has highlighted several key molecular pathways that may contribute to aging. These include pathways involved in DNA repair, inflammation, telomere maintenance, and cellular senescence. Identifying these pathways helps to understand the biological mechanisms underlying increased epigenetic age acceleration and aging itself.
Future Directions and Implications
– Personalized Medicine: Understanding the genetic factors that contribute to epigenetic age acceleration could lead to personalized interventions aimed at slowing the aging process and preventing age-related diseases.
– Lifestyle and Environmental Interactions: Beyond genetics, it’s crucial to study how lifestyle and environmental factors interact with genetic predispositions to influence epigenetic age acceleration. Such research could lead to comprehensive strategies for healthy aging.
– Therapeutic Targets: Identifying genetic variants and pathways associated with accelerated aging provides targets for therapeutic interventions. Drugs or lifestyle modifications that can modify these targets may slow aging and improve healthspan.
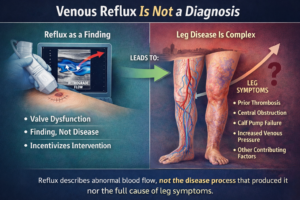
Aging and Vascular Health
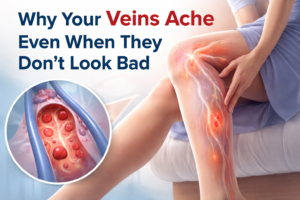
Aging and vascular health are intricately connected, with changes in the vascular system being both a cause and consequence of the aging process. The vascular system, which comprises arteries, veins, and capillaries, is crucial for delivering oxygen and nutrients to tissues and removing waste products. As we age, several changes occur in the vascular system that can lead to increased risk of cardiovascular diseases (CVDs), including hypertension, atherosclerosis, and peripheral artery disease. Understanding these changes and their impact on health is key to developing strategies for maintaining vascular health into older age.
Key Changes in Vascular Health with Aging
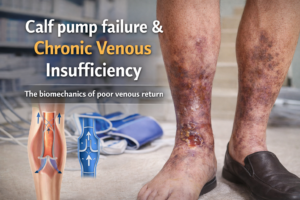
1. Endothelial Dysfunction: The endothelium is the inner lining of blood vessels and plays a critical role in vascular health by regulating blood flow and preventing thrombosis. Aging is associated with endothelial dysfunction, characterized by a reduction in the availability of nitric oxide (NO) and an increase in oxidative stress. This dysfunction can lead to impaired vasodilation, increased vascular stiffness, and a higher risk of atherosclerosis.
2. Increased Vascular Stiffness: With age, the walls of the arteries become stiffer due to structural changes, including collagen accumulation and elastin degradation. This increased stiffness can lead to higher systolic blood pressure and pulse pressure, straining the heart and increasing the risk of cardiovascular events.
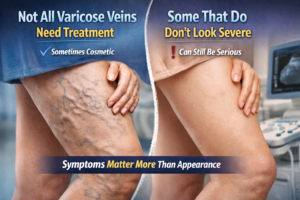
3. Atherosclerosis: Aging is a major risk factor for the development of atherosclerosis, a condition characterized by the buildup of plaques within the arterial walls. These plaques can narrow and harden arteries, restricting blood flow and potentially leading to heart attacks and strokes.
4. Inflammation: Chronic low-grade inflammation is a hallmark of aging, often referred to as “inflammaging.” This systemic inflammation can contribute to vascular damage and the progression of age-related vascular diseases.
Strategies for Maintaining Vascular Health
1. Healthy Lifestyle: Regular physical activity, a balanced diet rich in fruits, vegetables, and whole grains, and avoiding smoking can help maintain endothelial function and reduce the risk of vascular stiffness and atherosclerosis.
2. Blood Pressure Management: Managing blood pressure through lifestyle changes and medication when necessary is crucial for reducing the strain on blood vessels and preventing damage.
3. Cholesterol Management: Keeping cholesterol levels in check, particularly low-density lipoprotein (LDL) cholesterol, can help prevent the development and progression of atherosclerotic plaques.
4. Anti-inflammatory Interventions: Strategies to reduce chronic inflammation, including dietary supplements like omega-3 fatty acids, and medications, may help protect vascular health.
5. Screening and Monitoring: Regular health check-ups and monitoring of vascular health markers can help detect early signs of vascular aging, allowing for timely interventions.
Future Directions in Research
Research into aging and vascular health continues to explore the underlying mechanisms of vascular aging and the development of targeted interventions. Areas of interest include the potential of anti-aging therapies to improve vascular health, such as senolytics (drugs that target and eliminate senescent cells) and interventions aimed at enhancing endothelial function or reducing oxidative stress. Additionally, the role of genetic and epigenetic factors in vascular aging is an ongoing area of study, offering the possibility of personalized approaches to cardiovascular and metabolic health even in older age.
How can you improve your vascular health?
Improving vascular health is crucial for healthy longevity, reducing the risk of cardiovascular diseases and enhancing overall well-being. Here are several evidence-based strategies to improve your vascular health:
1. Maintain a Healthy Diet:
– Eat plenty of fruits and vegetables: These are rich in vitamins, minerals, and antioxidants that can help reduce inflammation and improve endothelial function.
– Choose whole grains over refined grains: Whole grains help in managing blood pressure and improving cholesterol levels.
– Include healthy fats: Foods high in omega-3 fatty acids, such as fish, nuts, and seeds, can decrease the risk of arterial plaques and lower blood pressure.
– Limit saturated fats and trans fats: Reducing intake of these fats can help lower your cholesterol levels and reduce plaque formation in your arteries.
– Stay hydrated: Drinking enough water is essential for maintaining the health of every cell in your body, including those in your vascular system.
2. Exercise Regularly:
– Engaging in regular physical activity, such as brisk walking, cycling, or swimming for at least 150 minutes per week, can improve heart and vascular health by lowering blood pressure, improving lipid profile, and enhancing endothelial function.
3. Maintain a Healthy Weight:
– Being overweight or obese increases the risk of vascular diseases. Losing weight can help reduce this risk and improve vascular function.
4. Quit Smoking:
– Smoking is a major risk factor for the development of vascular diseases. Quitting smoking can improve your vascular health significantly, as it reduces inflammation and improves endothelial function.
5. Manage Stress:
– Chronic stress can contribute to vascular damage over time. Techniques such as mindfulness, meditation, deep breathing exercises, and regular physical activity can help manage stress levels.
6. Limit Alcohol Consumption:
– While moderate alcohol consumption has been linked to some cardiovascular benefits, excessive drinking can damage the heart and blood vessels. Limiting alcohol intake can help protect vascular health.
7. Monitor and Manage Blood Pressure:
– High blood pressure (hypertension) is a leading cause of vascular damage. Regular monitoring and taking steps to manage high blood pressure through lifestyle changes and medication (if prescribed) are vital.
8. Control Blood Sugar Levels:
– High blood sugar levels can damage blood vessels over time, leading to vascular complications. Managing blood sugar levels through diet, exercise, and medication (if necessary) is crucial for individuals with diabetes or prediabetes.
9. Manage Cholesterol Levels:
– High levels of LDL (bad) cholesterol can lead to plaque buildup in the arteries, while high levels of HDL (good) cholesterol can help protect against heart disease. A healthy diet, regular exercise, and medication (if needed) can help manage cholesterol levels.
10. Regular Medical Check-ups:
– Regular check-ups with your healthcare provider can help detect and manage risk factors for vascular disease early on. This includes screening for high blood pressure, diabetes, and high cholesterol.
Implementing these strategies can lead to significant improvements in vascular health, reducing the risk of cardiovascular diseases and enhancing overall health and longevity.